Toxoplasmosis and intestinal protozoa
Research Lines
Content with Investigacion .
Trasplante de órganos
null
Presentación y Regulación Inmunes
null
Inmunología Microbiana e Inmunogenética
1. Análisis de la respuesta innata de mamíferos en la infección por Leishmania.
2. Caracterización inmunoproteómica en :
a. Streptococcus suis
b. Lactococcus garviae
c. Mycobacterium spp
3. Desarrollo de inmunoensayos analíticos basados en anticuerpos monoclonales (AcM) para detectar y cuantificar antígenos de origen animal, vegetal y microbiano.
4. Desarrollo y caracterización de AcM frente a los componentes del sistema del Complemento. Aplicación diagnóstica.
5. Desarrollo de reactivos de referencia y diseño de inmunoensayos para la evaluación cualitativa y cuantitativa de toxinas clostridiales.
6. Oferta tecnológica de producción de AcM y policlonales frente a substancias de interés industrial y biomédico.
El grupo está interesado en el estudio de la respuesta inmune desde una perspectiva multidisciplinar que incluye aproximaciones bioquímicas, biotecnológicas, genómicas, inmunoinformáticas y proteómicas, que junto con el uso adicional de modelos in vivo se encaminan al diseño de estrategias terapéuticas frente a diversas enfermedades crónicas, infecciosas y raras que poseen un claro componente inmunológico en su etiología.
Las principales líneas de investigación que está desarrollando el grupo en la actualidad son:
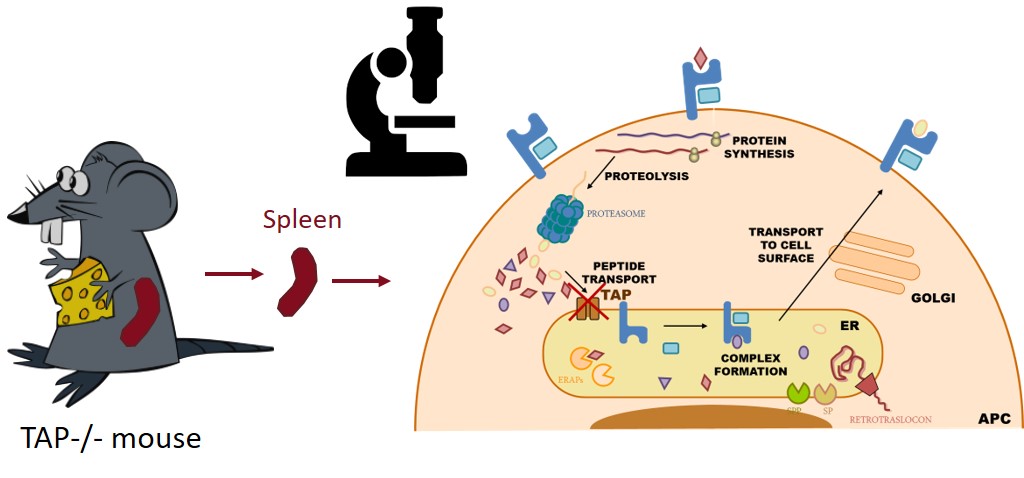
- * Análisis de las respuestas inmunes celulares frente a patógenos virales y bacterianos, mediante técnicas inmunoproteómicas, modelos in vivo con animales transgénicos y muestras humanas.
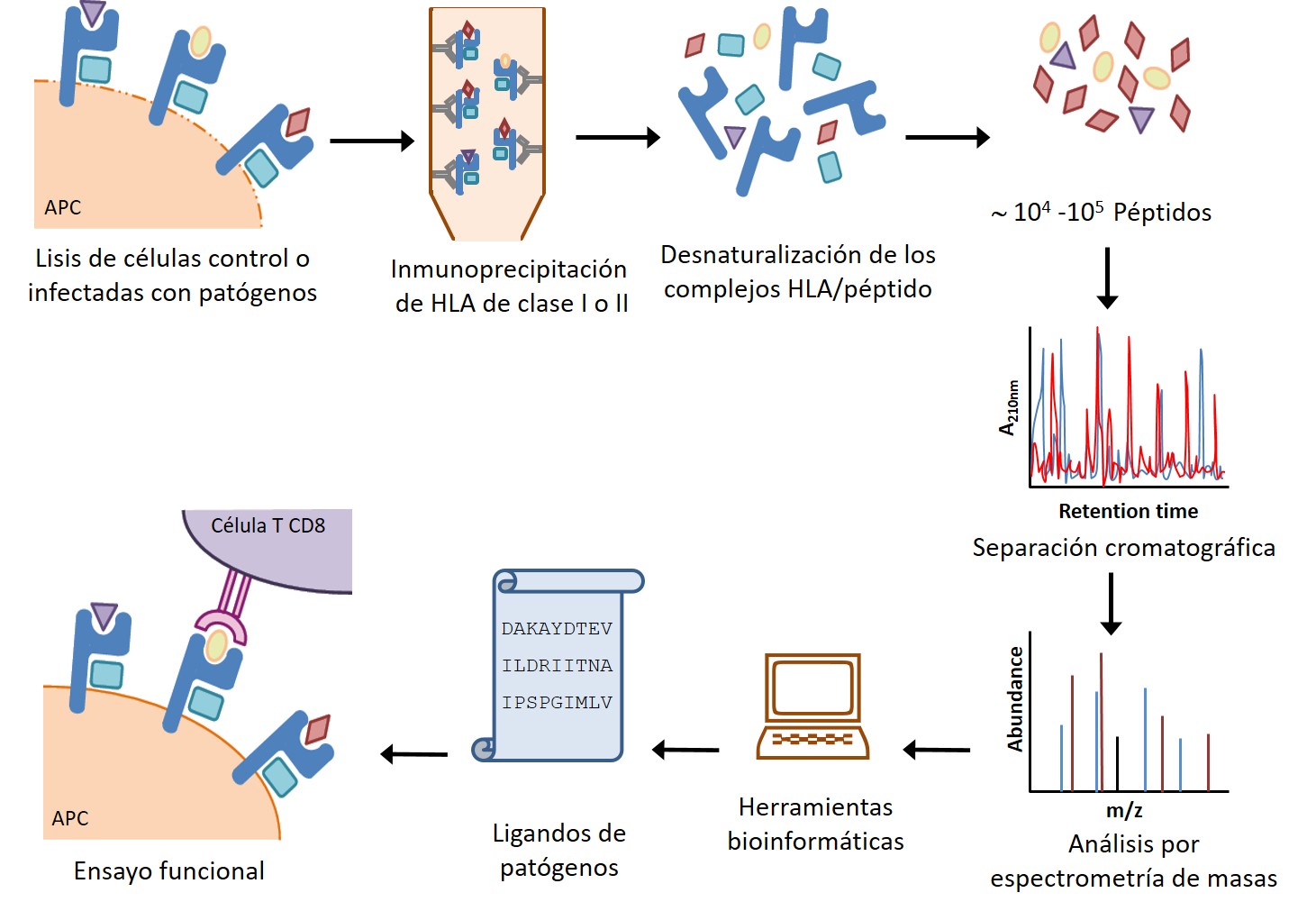
- * Caracterización de CD69: regulación génica, función reguladora inmune en homeostasis e infección y su uso como diana terapéutica, edición génica por CRISPR en modelos animales y celulares, etc.

* Desarrollo de herramientas inmunoinformáticas que permitan analizar la respuesta inmune celular frente a diversos virus de interés sanitario y determinar la eficacia de sus vacunas a nivel de población mundial.

* Estudio de las respuestas inmunes celulares frente a enfermedades raras (artritis reactiva y síndrome del linfocito desnudo) y crónicas (espondiloartropatías).
* Inclusión de componentes del sistema inmune en la fabricación de tejidos humanos, especialmente piel, para uso clínico, farmacéutico y cosmético.
- * Generación de virus recombinantes como vectores vacunales.
Inmunología Celular
null
The Immunobiology group has been working for years on the following lines of research:
1) The mechanisms of haematopoietic cell generation throughout ontogeny and the influence that the first haematopoietic cells exert on the innate and adaptive immune system present in the adults. We have identified and characterised a new population of B lymphocytes called B1-Rel (B220lo), which produce high levels of natural IgG/IgA antibodies. We sought to understand their role in the immune response in animal models of infection, analysing their impact on immune cell populations and on the production of soluble mediators (cytokines and immunoglobulins). In this regard, we have evaluated the generation of embryonic megakaryocytes (and their differentiation niches), their functionality and that of platelets, and their influence on haematopoietic development. For lymphoid populations, we have carried out extensive characterisation by flow cytometry and single cell RNA sequencing (scRNAseq) methodology. To carry out these cellomic studies, we have designed complex panels for use in multiparametric phenotypic analysis, and single cell cytometry and RNAseq omics technologies on purified cell populations.
In parallel, we are interested in understanding local immune responses in respiratory infections at times of particular susceptibility due to the fragility of the immune system (childhood and old age), both in mouse animal models, which allow their manipulation, and in humans.
2) Mouse models studied during neonatal life, in which we evaluated the effect of antibiotic (AB) treatment and addressed the role of TLR receptors in innate, pseudo-innate and adaptive immune cell populations. In these models, we observed that AB administration was able to modulate B-lymphoid populations, as well as their ability to secrete proinflammatory cytokines in culture and their differentiation into plasma cells, with differentiated immunoglobulin repertoires. Furthermore. These effects were mediated through the Toll-like receptor-2 (TLR2).
3) Mouse models with accelerated senescence (SAMP8) and senescent animals (over 20 months of age) to map lymphoid populations and soluble mediators of the immune response (immunoglobulins and cytokines). In these models, the B lymphoid populations (B1Rel and marginal zone B lymphocytes) are observed to be altered, accompanied by an increase in IgG1 with great restriction of their VDJ repertoires.
4) Role of the B1Rel population in animal models of local or systemic infection. We analysed the response to Streptoccoccus pneumoniae (SPN) locally in the lung and systemically in the spleen, as well as the role of TLR4 in these responses.
5) In humans, we are studying immune responses in children with respiratory syncytial virus (RSV) viral primo-infection. In this case we studied the immune response that occurs locally in the nasal mucosa (by analysis of nasal washings, NW) in a cohort of infected children versus healthy controls, stratified by age. We found that lymphomyeloid cells accumulate in these nasal washings in patients with diverse lymphocyte populations, as well as cytokines and immunoglobulins.
6) Analysis and characterisation of extracellular vesicles produced during respiratory infection both in lung supernatants from models of SPN infection and in LN in the case of children with RSV infection.
7) In parallel, we carry out studies of the genetic rearrangements of immunoglobulins and their use in the generation of chimeric receptors for possible use in immunotherapy.
Research projects
Content with Investigacion .
Los proyectos del grupo de los últimos años son los siguientes:
Proyecto “Enfoques inmunoinformaticos e inmunoproteomicos para identificar epitopos bacterianos implicados en la REA: diagnostico temprano y diseño de farmacos” financiado por el Plan Nacional de I+D+i del Ministerio de Ciencia, Innovación y Universidades. Centro Nacional de Microbiología, Instituto de Salud Carlos III. Investigador principal. Año: 2024-2026. Presupuesto Concedido: 225.000 euros. Proyecto PID2023-148729OB-100 financiado por MICIU/AEI/10.13039/501100011033 y por FEDER, UE.
Proyecto “La interrelación de CD69 y el procesamiento antigénico en enfermedades infecciosas y autoinmunes" financiado por la Acción Estratégica en Salud del Ministerio de Ciencia, Innovación y Universidades. Año: 2023-2025.
Proyecto “Interacciones génicas y proteicas de CD69 y sus regiones génicas reguladoras con moléculas" inanciado por el Plan Nacional de I+D+i del Ministerio de Ciencia, Innovación y Universidades. Centro Nacional de Microbiología, Instituto de Salud Carlos III. Proyecto PID2021-125757OB-100 financiado por MICIU/AEI/10.13039/501100011033 y por FEDER, UE.
Proyecto “Nuevas tecnologías de fabricación y optimización de tejidos: la piel como sistema modelo” financiado por el Programa de Actividades de I+D entre grupos de investigación de la Comunidad de Madrid en tecnologías 2018. Año: 2020-2023. Proyecto Coordinado por el Dr. Pablo Acedo de la Universidad Carlos III.
Proyecto “Estudio de CD69 como diana para mejorar el tratamiento de la leucopania y la movilización de células T de memoria de médula ósea" financiado por la Acción Estratégica en Salud del Ministerio de Ciencia, Innovación y Universidades. Año:2020-2024.
Proyecto “Diseño racional de una vacuna contra el virus respiratorio sincitial humano” financiado por la Acción Estratégica en Salud del Ministerio de Ciencia, Innovación y Universidades. Año: 2019-2022
Proyecto “Función de CD69 y sus elementos reguladores" financiado por la Acción Estratégica en Salud del Ministerio de Ciencia, Innovación y Universidades. Año: 2017-2022.
Proyecto “Diseño de vacunas recombinantes poliepitópicas para generar respuestas CD8+ contra virus emergentes” financiado por el Plan Nacional de I+D+i del Ministerio de Economía y Competitividad. Año: 2015-2017.
Proyecto “Análisis de los efectos de CD69 dependientes de S1P1 en modelos de infección e inflamación y estudio de su regulación” financiado por el FIS. Año: 2014-2017.
Proyecto “ADELVAC: Adenovirus con delecciones epitópicas para vacunación” financiado por el programa INNPACTO del Ministerio de Economía y Competitividad. Centro Nacional de Microbiología, Instituto de Salud Carlos III. Año: 2012-2014. Proyecto Coordinado por el Dr. Manel Cascallo de VCN BIOSCIENCES SL.
Proyecto “Diseño de vacunas multiepitópicas recombinantes para aumentar la respuesta inmune celular contra el VRSH” financiado por el Plan Nacional de I+D+i del Ministerio de Ciencia e Innovación. Año: 2012-2014.
-Project “Induction, differentiation and modulation of resident B lymphocytes in the lung in response to pneumococcus (NEUBLUNG)”. Ministry of Science and Innovation, PID2022-141754OB-I00 Call 2022 "Knowledge Generation Projects". 09/01/2023-08/31/2026. Financed by MICIU/AEI /10.13039/501100011033 and by ERDF, EU. PI: Belén by Andrés Muguruza. CoPI: María Luisa Gaspar Alonso-Vega.
-Project." Immune response of the nasal mucosa in childhood bronchiolitis” Instituto de Salud Carlos III-AESI. AESI-PI22CIII/00030 PI: Belén by Andrés Muguruza. CoPI Maria Luisa Gaspar Alonso-Vega. 01/01/2023-12/31/2025..
-Project. BenBedPhar. CA20121, European Union. Antonio Cuadrado. (CNM-ISCIII).10/19/2021-10/18/2025.
-Spanish Association Against Cancer Project “Novel comprehensive immunotherapy to specifically target the malignant clone in Sézary syndrome, an ultra-rare cancer of mature T lymphocytes”, number PROYE20084REGU. PI: José Ramón Regueiro, PI group Maria Luisa Gaspar. 01/01/2021-12/31/2023.
Project “The pulmonary immune system in homeostasis and infection: characterization and function of immature and pseudoinnate lymphoid populations.” MINECO-RETOS RTI2018-099114-B-100. PI: Maria Luisa Gaspar, CoPI: Belén de Andrés 01/01/2019-12/31/2022. Financed by MICIU/AEI /10.13039/501100011033/ and by FEDER A way of making Europe.
-Project “New B lymphoid populations: B1-rel pseudoinnate cells, homeostatic maintenance and their response under infection conditions.” MINECO-RETOS SAF2015-70880-R. PI: Maria Luisa Gaspar. 01/01/2016-12/31/2019.
-Project “Role of CD19+CD45R lymphocytes- in perinatal immune responses. Implications related to respiratory diseases in neonates. AESI PI14CIII/00049; PI Belén de Andrés. 2015-2018.
-Project “Study of the pseudo-innate population of CD19+CD45R- B lymphocytes in TLR-dependent infection models”. AESI PI11/01733FIS. PI Belén de Andrés. 2012-2015.
-Project." Cellular interactions in the establishment of B lymphoid differentiation niches: role of megakaryocytes and their implications in pathology. MINECO; SAF2012-33916. Maria Luisa Gaspar. 01/01/2013-12/31/2015.
-ISCIII Platforms Project to support R&D&I in Biomedicine and Health Sciences. PT23CIII/00006. 2023. Participating researcher: Isabel Cortegano.
-Research contracts between the Carlos III Health Institute and Inmunotek S.L. for the development of the Bactek-mv130 and Uromune-MV140 study in protection against S. pneumoniae infections. Immunotek. IP: Belen de Andrés 2019-2021.
-Research contract between the Carlos III Health Institute and Inmunotek S.L. “MV130 as a vaccine model based on trained immunity against respiratory infections due to pneumococcus and respiratory syncytial virus”, CAM Call. Industrial Doctorates. IND2023/BMD-27071. PI: Belén by Andrés Muguruza. 12/01/2023-11/30/2026.
- Titulo: “Inmunidad entrenada en trasplante de órganos”.
Entidad financiadora. Ministerio de Ciencia, Innovación y Universidades
Referencia: Proyecto PID2019-110015RB-I00 financiado por MICIU/AEI/10.13039/501100011033
IP: Jordi Cano Ochando
Fechas de ejecución: 01/06/2020-31/05/2024
Presupuesto: 205.700 €
Los proyectos del grupo de los últimos años son los siguientes:
Proyecto “La interrelación de CD69 y el procesamiento antigénico en enfermedades infecciosas y autoinmunes" financiado por la Acción Estratégica en Salud del Ministerio de Ciencia, Innovación y Universidades. Año: 2023-2025.
Proyecto “Interacciones génicas y proteicas de CD69 y sus regiones génicas reguladoras con moléculas" financiado por la AEI. Año: 2022-2024.
Proyecto “Nuevas tecnologías de fabricación y optimización de tejidos: la piel como sistema modelo” financiado por el Programa de Actividades de I+D entre grupos de investigación de la Comunidad de Madrid en tecnologías 2018. Año: 2020-2023. Proyecto Coordinado por el Dr. Pablo Acedo de la Universidad Carlos III.
Proyecto “Estudio de CD69 como diana para mejorar el tratamiento de la leucopania y la movilización de células T de memoria de médula ósea" financiado por la Acción Estratégica en Salud del Ministerio de Ciencia, Innovación y Universidades. Año:2020-2024.
Proyecto “Diseño racional de una vacuna contra el virus respiratorio sincitial humano” financiado por la Acción Estratégica en Salud del Ministerio de Ciencia, Innovación y Universidades. Año: 2019-2022
Proyecto “Función de CD69 y sus elementos reguladores" financiado por la Acción Estratégica en Salud del Ministerio de Ciencia, Innovación y Universidades. Año: 2017-2022.
Proyecto “Diseño de vacunas recombinantes poliepitópicas para generar respuestas CD8+ contra virus emergentes” financiado por el Plan Nacional de I+D+i del Ministerio de Economía y Competitividad. Año: 2015-2017.
Proyecto “Análisis de los efectos de CD69 dependientes de S1P1 en modelos de infección e inflamación y estudio de su regulación” financiado por el FIS. Año: 2014-2017.
Proyecto “ADELVAC: Adenovirus con delecciones epitópicas para vacunación” financiado por el programa INNPACTO del Ministerio de Economía y Competitividad. Centro Nacional de Microbiología, Instituto de Salud Carlos III. Año: 2012-2014. Proyecto Coordinado por el Dr. Manel Cascallo de VCN BIOSCIENCES SL.
Proyecto “Diseño de vacunas multiepitópicas recombinantes para aumentar la respuesta inmune celular contra el VRSH” financiado por el Plan Nacional de I+D+i del Ministerio de Ciencia e Innovación. Año: 2012-2014.
Publications
Viruses from a cluster of HIV-1 Elite controllers inherit viral characteristics associated with the clinical non-progressor phenotype
Casado C, Marrero-Hernández S, Márquez-Arce D, Pernas M, Marfil S, Borràs-Grañana F, Olivares I, Cabrera-Rodríguez R, Valera M-S, de Armas-Rillo L, Lemey P, Blanco J, Valenzuela-Fernández A, Lopez-Galíndez C. 2018. mBio 9:e02338-17.
PUBMED DOIPermanent control of HIV-1 pathogenesis in exceptional elite controllers: a model of spontaneous cure
Casado C, Galvez C, Pernas M, Tarancon-Diez L, Rodriguez C, Sanchez-Merino V, Vera M, Olivares I, De Pablo-Bernal R, Merino-Mansilla A, Del Romero J, Lorenzo-Redondo R, Ruiz-Mateos E, Salgado M, Martinez-Picado J, Lopez-Galindez C. Sci Rep. 2020 Feb 5,10(1):1902
PUBMED DOIHigh-Risk Sexual Practices Contribute to HIV-1 Double Infection Among Men Who Have Sex with Men in Madrid
Casado C, Pernas M, Rava M, Ayerdi O, Vera M, Alenda R, Jiménez P, Docando F, Olivares I, Zaballos A, Vicario JL, Rodríguez C, Del Romero J, Lopez-Galindez C. AIDS Res Hum Retroviruses. 2020 Nov, 36(11):896-904
PUBMED DOIIdentification of a Spanish HIV-1 Long Term Non-Progressor cluster infected with a low replicating virus
Concepción Casado, Maria Pernas, Virginia Sandonis, Tamara Alvaro-Cifuentes, Isabel Olivares, Rosa Fuentes, Lorena Martínez Prats, Eulalia Grau, Lidia Ruiz, Rafael Delgado, Carmen Rodríguez, Jorge del Romero, and Cecilio López-Galíndez. (2013). PLoS One. 8 (10):e77663.
PUBMED DOIImmunoescape of HIV-1 in Env-EL9 CD8 + T cell response restricted by HLA-B*14:02 in a Non progressor who lost twenty-seven years of HIV-1 control
Moyano A, Blanch-Lombarte O, Tarancon-Diez L, Pedreño-Lopez N, Arenas M, Alvaro T, Casado C, Olivares I, Vera M, Rodriguez C, Del Romero J, López-Galíndez C, Ruiz-Mateos E, Prado JG, Pernas M. Retrovirology. 2022 Mar 26,19(1):6
PUBMED DOIThe Characteristics of the HIV-1 Env Glycoprotein Are Linked with Viral Pathogenesis
Pérez-Yanes S, Pernas M, Marfil S, Cabrera-Rodríguez R, Ortiz R, Urrea V, Rovirosa C, Estévez-Herrera J, Olivares I, Casado C, Lopez-Galindez C, Blanco J, Valenzuela-Fernández A. Front Microbiol. 2022 Mar 24, 3:763039.
PUBMED DOIAnalysis of HIV-1 “in vitro” evolution through 3D real fitness landscapes constructed by Self Organizing Maps.
Ramón Lorenzo‐Redondo, Soledad Delgado, Federico Morán, and Cecilio Lopez‐Galindez. Analysis of HIV-1 “in vitro” evolution through 3D real fitness landscapes constructed by Self Organizing Maps. (2014) PLoS One. 9(2): e88579.
PUBMED DOIPhenotypic and molecular characterization of IMP-producing Enterobacterales in Spain: Predominance of IMP-8 in Klebsiella pneumoniae and IMP-22 in Enterobacter roggenkampii.
5. Phenotypic and molecular characterization of IMP-producing Enterobacterales in Spain: Predominance of IMP-8 in Klebsiella pneumoniae and IMP-22 in Enterobacter roggenkampii. Autores: Cañada-García JE, Grippo N, de Arellano ER, Bautista V, Lara N, Navarro AM, Cabezas T, Martínez-Ramírez NM, García-Cobos S, Calvo J, Cercenado E, Aracil B, Pérez-Vázquez M, Oteo-Iglesias J; Spanish IMP Study Group. Revista: Front Microbiol. 2022 Sep 28;13:1000787.
DOIHCV eradication with DAAs differently affects HIV males and females: A whole miRNA sequencing characterization
Valle-Millares D; Brochado-Kith O; Gómez-Sanz A; et al; Fernández-Rodríguez A (AC). (17/17). 2021. Biomedicine and Pharmacotherapy. Elsevier.
DOIOutbreak of invasive meningococcal disease caused by a meningococcus serogroup B expressing a rare porA genosubtype (19-54, 15), Spain, March to April 2024.
Abad R, Navarro C, García-Amil C, Montes M, Castañeda-García A, Cuadros JA, Galar A, Martin F, Mena E, Pérez de Madrid S, Román C, Soler M, Vázquez JA. Euro Surveill. 2025 Nov;30(44):2500222
PUBMED DOIT-Cell-Specific Loss of the PI-3-Kinase p110α Catalytic Subunit Results in Enhanced Cytokine Production and Antitumor Response.
1. Aragoneses-Fenoll L, Ojeda G, Montes-Casado M, Acosta-Ampudia Y, Dianzani U, Portolés P, Rojo JM. T-Cell-Specific Loss of the PI-3-Kinase p110α Catalytic Subunit Results in Enhanced Cytokine Production and Antitumor Response. Front. Immunol. 2018 Feb 27;9:332.
PUBMED DOIEssential in vitro diagnostics for advanced HIV and serious fungal diseases: international experts' consensus recommendations. Eur J Clin Microbiol Infect Dis. 2019 Sep
Bongomin F, Govender NP, Chakrabarti A, Robert-Gangneux F, Boulware DR, Zafar A, Oladele RO, Richardson MD, Gangneux JP, Alastruey-Izquierdo A, Bazira J, Boyles TH, Sarcarlal J, Nacher M, Obayashi T, Worodria W, Pasqualotto AC, Meya DB, Cheng B, Sriruttan C, Muzoora C, Kambugu A, Rodriguez Tudela JL, Jordan A, Chiller TM, Denning DW. Essential in vitro diagnostics for advanced HIV and serious fungal diseases: international experts' consensus recommendations. Eur J Clin Microbiol Infect Dis. 2019 Sep;38(9):1581-1584. doi: 10.1007/s10096-019-03600-4. PMID: 31175479.
PUBMED DOIPneumoviridae fusion proteins as immunogens to induce cross-neutralizing antibody responses
Olmedillas E, Cano O, Martinez I, Luque D, Terron MC, McLellan JS, et al. Chimeric Pneumoviridae fusion proteins as immunogens to induce cross-neutralizing antibody responses. EMBO Mol Med. 2018;10(2):175-87.
PUBMED DOIHigh-Quality Draft Genome of Babesia divergens, the Etiological Agent of Cattle and Human Babesiosis.
8. Cuesta I; González LM; Estrada K; Grande R; Zaballos A; Lobo CA; Barrera J; Sanchez-Flores A; Montero E. 2014. High-Quality Draft Genome of Babesia divergens, the Etiological Agent of Cattle and Human Babesiosis. Genome Announcement. 2: e01194-14.
PUBMED DOIEmergence of linezolid-resistant coagulase-negative staphylococci in an intensive care unit.
Emergence of linezolid-resistant coagulase-negative staphylococci in an intensive care unit. Balandin B, Lobo B, Orden B, Román F, García E, Martínez R, Valdivia M, Ortega A, Fernández I, Galdos P. Infect Dis (Lond). 2016;48(5):343-9.
PUBMEDCo-occurrence of the cephalosporinase cepA and carbapenemase cfiA genes in a Bacteroides fragilis division II strain, an unexpected finding
Valdezate S, Medina-Pascual MJ, Villalón P, Garrido N, Monzón S, Cuesta I, Cobo F (2024). Co-occurrence of the cephalosporinase cepA and carbapenemase cfiA genes in a Bacteroides fragilis division II strain, an unexpected finding. J Antimicrobial Chem. 2024 Jul 1;79(7):1683-1687
PUBMED DOIHookworm secreted extracellular vesicles interact with host cells and prevent inducible colitis in mice.
Eichenberger RM, Ryan S, Jones L, Buitrago G, Polster R, Montes de Oca M, Zuvelek J, Giacomin PR, Dent LA, Engwerda CR, Field MA, Sotillo J, Loukas A. Hookworm secreted extracellular vesicles interact with host cells and prevent inducible colitis in mice. Front Immunol. 2018 Apr 30;9:850.
PUBMED DOIToll-like receptor signaling-deficient cells enhance antitumor activity of cell-based immunotherapy by increasing tumor homing
A. Morales-Molina, M.A. Rodríguez-Milla, S,. Gambera, T. Cejalvo, B. de Andrés M.L. Gaspar, J. Garcia-Castro. Cancer Res Commun 2023 Mar 1;3(3):347-360. eCollection 2023 Mar
PUBMED DOICharacterizing carbapenemase-producing Escherichia coli isolates from Spain: high genetic heterogeneity and wide geographical spread.
1. Characterizing carbapenemase-producing Escherichia coli isolates from Spain: high genetic heterogeneity and wide geographical spread. Dahdouh E, Gómez-Marcos L, Cañada-García JE, de Arellano ER, Sánchez-García A, Sánchez-Romero I, López-Urrutia L, de la Iglesia P, Gonzalez-Praetorius A, Sotelo J, Valle-Millares D, Alonso-González I, Bautista V, Lara N, García-Cobos S, Cercenado E, Aracil B, Oteo-Iglesias J, Pérez-Vázquez M; Spanish Eco-Carba Study Group. Revista: Front Cell Infect Microbiol 2024 May 16;14:1390966.
PUBMED DOIThe sulfur-related metabolic status of Aspergillus fumigatus during infection reveals cytosolic serine hydroxymethyltransferase as a promising antifungal target
Alharthi R, Sueiro-Olivares M, Storer I, Bin Shuraym H, Scott J, Al-Shidhani R, Fortune-Grant R, Bignell E, Tabernero L, Bromley M and Amich J. 2025. Virulence, 16(1):2449075
PUBMED DOIThe importance of microbiology reference laboratories and adequate funding for infectious disease surveillance
Shaw D, Torreblanca RA, Amin-Chowdhury Z, Bautista A, Bennett D, Broughton K, Casanova C, Choi EH, Claus H, Corcoran M, Cottrell S, Cunney R, Cuypers L, Dalby T, Davies H, de Gouveia L, Deghmane AE, Desmet S, Domenech M, Drew R, Plessis MD, Duarte C, Fuursted K, Golden A, Almeida SCG, Henares D, Henriques-Normark B, Hilty M, Hoffmann S, Humphreys H, Jacobsson S, Johnson C, Jolley KA, Kawabata A, Kozakova J, Kristinsson KG, Krizova P, Kuch A, Ladhani S, Lâm TT, Ayala MEL, Lindholm L, Litt D, Maiden MCJ, Martin I, Martiny D, Mattheus W, McCarthy ND, Meehan M, Meiring S, Mölling P, Morfeldt E, Morgan J, Mulhall R, Muñoz-Almagro C, Murdoch D, Musilek M, Novakova L, Oftadeh S, Perez-Arguello A, Pérez-Vázquez MD, Perrin M, Prevost B, Roberts M, Rokney A, Ron M, Sanabria OM, Scott KJ, Sempere J, Siira L, de Lemos APS, Sintchenko V, Skoczyńska A, Slotved HC, Smith AJ, Taha MK, Toropainen M, Tzanakaki G, Vainio A, van der Linden MPG, van Sorge NM, Varon E, Moreno JV, Vohrnova S, von Gottberg A, Yuste J, Brueggemann AB. Lancet Digit Health. 2025 Apr;7(4):e275-e281.
PUBMED DOIETP-46321, a dual p110α/δ class IA phosphoinositide 3-kinase inhibitor modulates T lymphocyte activation and collagen-induced arthritis.
2. Aragoneses-Fenoll L, Montes-CasadoM, Ojeda G, Acosta YY, Herranz J, Martínez S, Blanco-Aparicio C, Criado G, Pastor J, Dianzani U, Portolés P, Rojo JM. ETP-46321, a dual p110α/δ class IA phosphoinositide 3-kinase inhibitor modulates T lymphocyte activation and collagen-induced arthritis. Biochem. Pharmacol. 2016 Apr 15;106:56-69. Epub 2016 Feb 13.
PUBMED DOIStructure and Immunogenicity of the Human Metapneumovirus F Protein in the Postfusion Conformation.
5. Mas V, Rodriguez L, Olmedillas E, Cano O, Palomo C, Terron MC, et al. Engineering, Structure and Immunogenicity of the Human Metapneumovirus F Protein in the Postfusion Conformation. PLoS Pathog. 2016;12(9):e1005859.
PUBMED DOIGodet C, Alastruey-Izquierdo A, Flick H, Hennequin C, Mikilps-Mikgelbs R, Munteanu O, Page I, Seidel D, Salzer HJF. A CPAnet consensus statement on research priorities for chronic pulmonary aspergillosis: a neglected fungal infection that requires attention. J Antimicrob Chemother. 2018
Godet C, Alastruey-Izquierdo A, Flick H, Hennequin C, Mikilps-Mikgelbs R, Munteanu O, Page I, Seidel D, Salzer HJF. A CPAnet consensus statement on research priorities for chronic pulmonary aspergillosis: a neglected fungal infection that requires attention. J Antimicrob Chemother. 2018 Feb 1;73(2):280-286. doi: 10.1093/jac/dkx390. PMID: 29126309.
PUBMED DOIExploring the genetic background of the botulism neurotoxin BoNT/B2 in Spain
Valdezate S, Carrasco G, Medina MJ, Garrido N, Del Pino S, Valiente M, Pallarés MP, Villalon P. (2023). Exploring the genetic background of the botulism neurotoxin BoNT/B2 in Spain. Microbiol Spectr. Sep 26;11(5):e0238023
PUBMED DOIHDP2: a ribosomal DNA (NTS-ETS) sequence as a target for species-specific molecular diagnosis of intestinal taeniasis in humans.
Flores MD, Gonzalez LM, Hurtado C, Motta YM, Domínguez-Hidalgo C, Merino FJ, Perteguer MJ, Gárate T. HDP2: a ribosomal DNA (NTS-ETS) sequence as a target for species-specific molecular diagnosis of intestinal taeniasis in humans. Parasit Vectors. 2018 Feb 27;11(1):117. doi: 10.1186/s13071-018-2646-6.
PUBMED DOIImmune stress suppresses innate immune signaling in preleukemic precursor B-cells to provoke leukemia in predisposed mice
Isidro-Hernández M, Casado-García A, Oak N, Alemán-Arteaga S, Ruiz-Corzo B, Martínez-Cano J, Mayado A, G. Sánchez E, Blanco O, Gaspar ML, Orfao A, Alonso-López D, De las Rivas J, Riesco S, Prieto-Matos P, González-Murilo A, García Criado FJ, García Cenador MB, Ramírez-Orellana M, De Andrés B, Vicente-Dueñas C, Cobaleda C, Nichols KE, Sánchez-García I. Nat Commun 2023 Aug 24;14(1):5159.
PUBMED DOIClinical, microbiological, and molecular characterization of pediatric invasive infections by Streptococcus pyogenes in Spain in a context of global outbreak.
2. Clinical, microbiological, and molecular characterization of pediatric invasive infections by Streptococcus pyogenes in Spain in a context of global outbreak. Autores: Ramírez de Arellano E, Saavedra-Lozano J, Villalón P, Jové-Blanco A, Grandioso D, Sotelo J, Gamell A, González-López JJ, Cervantes E, Gónzalez MJ, Rello-Saltor V, Esteva C, Sanz-Santaeufemia F, Yagüe G, Manzanares Á, Brañas P, Ruiz de Gopegui E, Carrasco-Colom J, García F, Cercenado E, Mellado I, Del Castillo E, Pérez-Vazquez M, Oteo-Iglesias J, Calvo C; Spanish PedGAS-Net/CIBERINFEC GAS Study Group. Revista: mSphere. 2024 Mar 26;9(3):e0072923.
PUBMED DOIPotent Induction of Envelope-Specific Antibody Responses by Virus-Like Particle Immunogens Based on HIV-1 Envelopes from Patients with Early Broadly Neutralizing Responses
Beltran-Pavez C, Bontjer I, Gonzalez N, Pernas M, Merino-Mansilla A, Olvera A, Miro JM, Brander C, Alcami J, Sanders RW, Sanchez-Merino V, Yuste E; J Virol. 2022 Jan 12;96(1):e0134321.
PUBMED DOIFungal burden assessment in hospital zones with different protection degrees
García-Gutiérrez L, Baena Rojas B, Ruiz M, Hernández Egido S, Ruiz-Gaitán AC, Laiz L, Pemán J, Cuétara-García MS, Mellado E & Martin-Sanchez PM. Build Environ, Volume 269, 1 February 2025, 112454
DOIAntimicrobial resistance and epidemiological aspects of Neisseria gonorrhoeae in the province of Lleida, Spain (2017-2024).
Cumplido A, Aramburu J, Font M, Montes M, Abad R, López E, Bernet A, Mormeneo S, Prats I, García M, Sánchez E, Bellés A. Enferm Infecc Microbiol Clin (Engl Ed). 2025 Mar;43(3):156-161.
PUBMED DOIContent with Investigacion .
-

María Luisa Gaspar Alonso-Vega
Research Professor
ORCID code: 0000-0001-9858-3862
Dr. María Luis Gaspar Alons-Vega graduated in 1980 and obtained her PhD in 1985 in Medicine and Surgery from the Autonomous University of Madrid. She completed the specialty of Immunology (1981-1985), and her doctoral thesis under the direction of Dr. Carmen Gutierrez, in the Immunology laboratory of the Puerta de Hierro Clinic directed by Dr. Miguel Kreisler. She completed a predoctoral stay in the Cytogenetics Laboratory of the National Institute of Autoimmune, Diabetes, Digestive and Kidney Diseases (NIDDK, NIH), under the supervision of Dr. JH Tjio and Dr. E. Raveché. She joined the Immunology Service of the National Center for Health Microbiology, Virology and Immunology (CNMVIS, AISNA and later ISCIII) as a Physician-Specialist in 1986, in the Immunology Laboratory directed by Dr. Alfredo Toraño. She completed a postdoctoral stay (1989-1991) at the Immunogenetics Unit of the Pasteur Institute (Paris) directed by Dr. T. Meo. From 1991 to 2006 she was Head of the Immunology Section successively at the CNMVIS, at the National Center for Fundamental Biology (CNBF-ISCIII) and at the National Center for Microbiology (CNM-ISCIII). From 2006 to 2016 she has been a Senior Researcher and Senior Scientist of OPIs, in the Immunobiology laboratory of the CNM-ISCIII. From 2016 to 2018 she was a Scientific Researcher at OPIs and since 2018, she is a Research Professor at OPIs at the CNM.
-

Belén de Andrés Muguruza
Research Scientist
ORCID code: 0000-0002-7391-2823
Graduated in Biology in 1987 and PhD in 1992 from the Autonomous University of Madrid. He completed his doctoral thesis in the laboratory of Dr. Carlos Lahoz in the Immunology department of the Jiménez Díaz Foundation with a pre-doctoral stay at the Institute Curie in Paris, in the laboratory of Dr. Wolf H. Fridman. Subsequently, he completed a two-year postdoctoral stay in the Department of Pathology of the College of Medicine at the University of Iowa, USA, in the laboratory of Dr. Richard G. Lynch. After a year as an Adjunct in the Immunology department of the Jiménez Diaz Foundation, she worked for 2 years with a reinstatement contract from the Ministry of Science in the Immunobiology department of the CNM/ISCIII in the laboratory of Dr. Mª Luisa Gaspar and later with a Ramón y Cajal contract. In 2006 she obtained a position as Staff Senior Scientist.
-

Isabel Cortegano Jimeno
Research Scientist
ORCID code: 0000-0002-6504-6347
Graduated in Biology in 1995 (Specialty in Biochemistry and Molecular Biology) and PhD in 1999 from the Autonomous University of Madrid. He completed his doctoral thesis at the Jiménez Díaz Foundation in the Immunology laboratory directed by Dr. Carlos Lahoz. Later he obtained a postdoctoral fellowship in the Immunobiology laboratory of Dr. Mª Luisa Gaspar at the National Center of Microbiology (CNM) of the Carlos IIII Health Institute (ISCIII) (2002-2006). He then enjoyed an I3P contract from the CSIC in the laboratory of Professor Miguel Ángel Rodríguez-Marcos (2007-2009). She has been a researcher associated with research projects in the ISCIII Immunobiology laboratory during the years 2010-2018. Since 2018 she has been an associate professor in the Department of Cell Biology of the UCM Faculty of Medicine. He coordinates the scientific dissemination group of the Spanish Society of Immunology (GESEI), is part of the editorial committee of the SEI magazine and is a member of the board of the CAM Immunology Society. She is a Senior Scientist of the ISCIII at the National Center for Microbiology since 2020.
-

Carolina Ruiz Sánchez
Specialized Technician
ORCID code: 0000-0002-2177-8132
Graduated in Chemical Sciences (biochemistry specialty) in 1998 and PhD in 2022 from the Complutense University of Madrid. In 2008 he joined the OPIS Assistant in the Immunobiology laboratory of the National Center for Microbiology of the ISCIII, specializing in flow cytometry the first year and subsequently becoming part of the laboratory's technical team. In 2012 he was promoted to Intermediate Level Technician and in 2018 to Higher Specialized Technician of the OPIS, currently occupying this position in the same laboratory.
-

Mercedes Rodríguez García
Specialized Higher Technician
Graduated in Biology from the Complutense University of Madrid in 2003, she began her doctoral studies in the Bone Metabolism laboratory of the La Paz University Hospital.
In 2007 he joined as a Research Assistant at the Carlos III Health Institute, at the CNM, in the Transplant Immunology laboratory. After 5 years, he began working in the Immunobilology laboratory where he was promoted in 2018 to Specialized Technician, in 2024 to Senior Specialized Technician and where he currently continues to develop his professional career. -

Alejandro Arrabal Sierra
Predoctoral Contract (Industrial Doctorate from CAM / Inmunotek).
ORCID code: 0000-0002-9354-9224
Graduated in Biotechnology in 2021 from the Polytechnic University of Madrid, carrying out his final degree project in the Immunobiology laboratory of the National Microbiology Center of the ISCIII. In 2021, he completed a Master's Degree in Research in Immunology at the Complutense University of Madrid and completed his master's thesis in the same CNM laboratory. Subsequently, he worked for a year as a Research Assistant in the laboratory of Dr. Elena Fernández Ruiz at the Hospital Universitario de la Princesa. Since the end of 2023, he has been a predoctoral fellow in the CNM Immunobiology laboratory with an Industrial Doctorate scholarship from the Community of Madrid in collaboration with the company Inmunotek.
-
Pilar Portolés Pérez
Científico Titular de OPIs. Jefa de Unidad
-
Begoña Galocha Iragüen
Científico Titular
-
Jordi Cano Ochando
Investigador principal
-
Patricia Conde Sanromán
Postdoctoral
-
Haisul Chang
Postdoctoral
-
María González Gómez
Estudiante de doctorado
-
Ana Belén Martin Ezquerra
Técnico especializada de OPIs
-
Soledad Crespo Carrasco
Técnico Contratada
-
Pilar Lauzurica Gómez
Investigador Principal
-
Daniel López Rodríguez
Investigador Principal
-
Elena Lorente Galán
Técnico Especializado de OPIS
-
Almudena Albentosa Cocho
Ayudante de Investigación
-
Carmen Mir Guerrero
Técnico de Laboratorio
-
Miguel Gómez Fontela
Contrato Predoctoral
List of staff
Additional Information
Our group carries out research studies in the diagnosis, reference and epidemiology of zoonoses and emerging diseases, both indigenous and imported, caused by protozoa. Coordinates the study in the human field with the relevance of the animal field and the environment (One Health initiative), with special interest in Toxoplasmosis, a highly prevalent zoonosis (WHO lists it as the 3rd food-borne zoonosis in Europe), presents a complex epidemiological cycle and causes neurological, ocular and systemic symptoms. We carry out diagnostic and characterization studies of Toxoplasma gondii from human and animal cases, to obtain greater epidemiological information and analyze the possible relationship with virulence and pathology.
Cryptosporidium, Giardia, Blastocystis and Entamoeba histolytica cause gastrointestinal diseases, affecting children, immunosuppressed people and travelers. They can cause outbreaks. We develop diagnostic and characterization studies of isolates from humans and animals, from different areas and countries, to establish the presence of the main species and genotypes and the epidemiological situation. We are beginning the study of associations between these parasites and the intestinal microbiota.
The pathogenic Free-Living Amoebas, Acanthamoeba, Naegleria fowleri and Balamuthia mandrillaris, cause emerging diseases, highlighting the importance of the environment in transmission. They cause underdiagnosed neurological and ocular cases. The diagnostic and genotyping study of human and animal isolates that we are carrying out aims to establish the real prevalence, transmission routes and epidemiology.
Our group carries out research studies in the diagnosis, reference and epidemiology of zoonoses and emerging diseases, both indigenous and imported, caused by protozoa. Coordinates the study in the human field with the relevance of the animal field and the environment (One Health initiative), with special interest in Toxoplasmosis, a highly prevalent zoonosis (WHO lists it as the 3rd food-borne zoonosis in Europe), presents a complex epidemiological cycle and causes neurological, ocular and systemic symptoms. We carry out diagnostic and characterization studies of Toxoplasma gondii from human and animal cases, to obtain greater epidemiological information and analyze the possible relationship with virulence and pathology.
Cryptosporidium, Giardia, Blastocystis and Entamoeba histolytica cause gastrointestinal diseases, affecting children, immunosuppressed people and travelers. They can cause outbreaks. We develop diagnostic and characterization studies of isolates from humans and animals, from different areas and countries, to establish the presence of the main species and genotypes and the epidemiological situation. We are beginning the study of associations between these parasites and the intestinal microbiota.
The pathogenic Free-Living Amoebas, Acanthamoeba, Naegleria fowleri and Balamuthia mandrillaris, cause emerging diseases, highlighting the importance of the environment in transmission. They cause underdiagnosed neurological and ocular cases. The diagnostic and genotyping study of human and animal isolates that we are carrying out aims to establish the real prevalence, transmission routes and epidemiology.